Why Biological Age Matters
Biological age quantifies the functional health of cells, tissues, and organ systems, whereas chronological age simply records the number of years lived. Modern epigenetic clocks (e.g., DunedinPACE, Horvath’s DNAmAge) and multi‑biomarker panels (clinical chemistry, inflammatory markers, telomere length) translate molecular damage into an age estimate that predicts energy capacity, immune competence, and disease risk more accurately than calendar age. A younger biological age correlates with higher mitochondrial ATP production, robust naïve T‑cell repertoires, and lower systemic inflammation, translating into better daily vigor and reduced incidence of cardiovascular disease, diabetes, and neurodegeneration. Advances in DNA‑methylation profiling, proteomics, and metabolomics now allow clinicians to track biological age longitudinally, identify organ‑specific aging gaps, and tailor preventive interventions—diet, exercise, sleep, and emerging therapeutics—to compress morbidity and extend healthspan.
Reversing Biological Age – The Core Strategy
8‑Week Reversal Protocol
| Component | Details | Reported DNAmAge Reduction |
|---|---|---|
| Nutrition | Folate‑rich greens, betaine (beets), vitamin C, vitamin A, polyphenols (curcumin, EGCG, quercetin) | 4.6 years average (range 1.2–11) |
| Sleep | 7–9 h nightly, consistent schedule | Contributes ~1–2 years reduction |
| Aerobic Exercise | Daily moderate‑intensity (≥30 min) | Improves mitochondrial biogenesis |
| Resistance Training | Bi‑weekly sessions (≥2 × week) | Preserves muscle mass, adds ~0.5 yr benefit |
| Stress Management | Twice‑daily breathing exercises, mindfulness | Reverses acute stress spikes |
| Population | Women, 8‑week trial | Average 4.6 yr reduction in Horvath DNAmAge |
 Advances in DNA‑methylation clocks (e.g., Horvath, DunedinPACE) show that biological age is a mutable metric. Epigenetic remodeling can be driven by nutrition, exercise, sleep and stress management, yielding measurable reductions in DNAmAge.
Advances in DNA‑methylation clocks (e.g., Horvath, DunedinPACE) show that biological age is a mutable metric. Epigenetic remodeling can be driven by nutrition, exercise, sleep and stress management, yielding measurable reductions in DNAmAge.
How to reverse biological age – An 8‑week protocol that supplies folate‑rich greens, betaine from beets, vitamin C, vitamin A and polyphenols (curcumin, EGCG, quercetin) while enforcing 7‑9 h nightly sleep, daily moderate‑intensity aerobic activity, bi‑weekly resistance training and twice‑daily breathing exercises lowered Horvath DNAmAge by an average of 4.6 years in women (range 1.2–11 years). Acute stressors (surgery, severe infection) transiently raise epigenetic age but return to baseline after recovery, highlighting the importance of stress‑reduction techniques.
Can you reverse biological age – Yes. Small case series and 8‑week lifestyle trials consistently demonstrate 2‑5‑year epigenetic age declines, and stress‑induced spikes are reversible. The magnitude varies; larger, long‑term studies are needed to refine protocols.
Is it possible to decrease your biological age? – Steady lifestyle changes—regular aerobic/strength exercise, Mediterranean‑style plant‑forward diet, adequate sleep, mindfulness‑based stress reduction, smoking cessation and optimal vitamin D—can modestly lower biological age and improve healthspan.
What does BioAge do? – BioAge Labs translates multi‑omics insights into therapeutics that target aging hallmarks (e.g., inflammation, mitochondrial dysfunction, immunosenescence), aiming to lower biological age pharmacologically and complement lifestyle interventions.
Dietary Protocols That Turn Back the Clock
Key Dietary Strategies
| Strategy | Core Nutrients | Typical Duration | Approx. Epigenetic Benefit |
|---|---|---|---|
| Whole‑Food Methylation‑Supportive Diet | Folate, B‑12, choline, omega‑3, antioxidants, phytonutrients | 8 weeks | 4–5 yr reduction (up to 11 yr) |
| Fasting‑Mimicking (5‑day/month) | Low‑calorie, plant‑focused | Monthly 5 days | ~2.5 yr reduction |
| Time‑Restricted Eating | 12‑hour overnight fast | Daily | Supports circadian methylation, modest benefit |
| Probiotic Timing | Lactobacillus plantarum 299v + greens powder (betaine, curcumin, EGCG) | Daily | Sustains DNA‑MTase activity, amplifies diet effect |
 Methylation‑supportive eating plans focus on whole‑food sources of folate, B‑12, choline, omega-3s, antioxidants and phytonutrients—leafy greens, berries, nuts, fatty fish, fermented foods and high‑quality protein. An eight‑week protocol that emphasizes these nutrients lowered DNA‑methylation‑based biological age by an average of 4–5 years in women, with some participants achieving up to an 11‑year reduction.
Methylation‑supportive eating plans focus on whole‑food sources of folate, B‑12, choline, omega-3s, antioxidants and phytonutrients—leafy greens, berries, nuts, fatty fish, fermented foods and high‑quality protein. An eight‑week protocol that emphasizes these nutrients lowered DNA‑methylation‑based biological age by an average of 4–5 years in women, with some participants achieving up to an 11‑year reduction.
Fasting‑mimicking and intermittent fasting add a metabolic stress that activates Nrf2 signaling, promotes mitochondrial biogenesis and improves insulin sensitivity. A monthly five‑day low‑calorie, plant‑focused fasting‑mimicking regimen further reduced epigenetic aging markers by about 2½ years, while daily time‑restricted eating (12‑hour overnight fast) supports circadian‑driven methylation patterns.
Probiotic and phytonutrient timing enhances gut‑derived methyl donors and anti‑inflammatory metabolites. Daily intake of Lactobacillus plantarum 299v or similar strains, combined with greens powders rich in betaine, curcumin and EGCG, sustains DNA‑methyltransferase activity and attenuates inflammation, amplifying the epigenetic benefits of diet and fasting.
Together, these evidence‑based dietary strategies—nutrient‑dense, methylation‑supportive foods, structured fasting, and targeted probiotic/phytonutrient timing—provide a personalized, preventive approach to “rewind” biological age and extend healthspan.
Exercise, Sleep, and Stress – The Triple Powerhouse
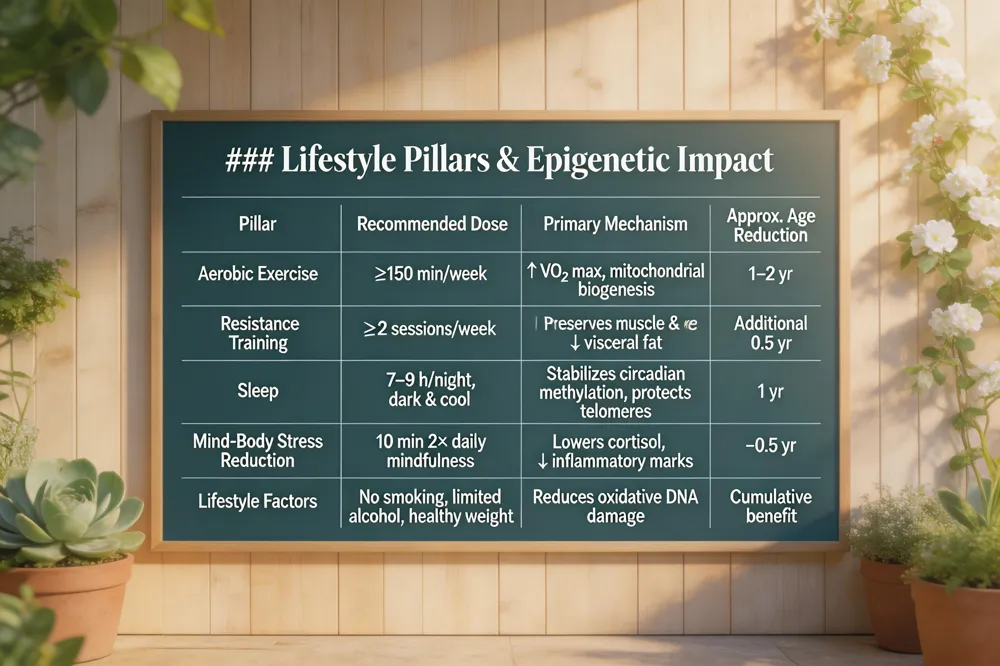
Lifestyle Pillars & Epigenetic Impact
| Pillar | Recommended Dose | Primary Mechanism | Approx. Age Reduction |
|---|---|---|---|
| Aerobic Exercise | ≥150 min/week | ↑ VO₂ max, mitochondrial biogenesis | 1–2 yr |
| Resistance Training | ≥2 sessions/week | Preserves muscle & bone, ↓ visceral fat | Additional 0.5 yr |
| Sleep | 7–9 h/night, dark & cool environment | Stabilizes methylation of circadian genes, protects telomeres | 1 yr |
| Mind‑Body Stress Reduction | Daily mindfulness/breathing (10 min 2×) | Lowers cortisol, ↓ inflammatory epigenetic marks | ~0.5 yr |
| Lifestyle Factors (no smoking, limited alcohol, healthy weight) | Ongoing | Reduces oxidative DNA damage | Cumulative benefit |
 9 powerful ways to reverse aging – Regular aerobic exercise (≥150 min/week) improves VO₂ max, mitochondrial biogenesis and lowers epigenetic age by 1–2 years. Adding resistance training (2 sessions/week) preserves muscle mass, bone density and visceral fat, further reducing biological age markers. A Mediterranean‑style diet rich in omega‑3s, polyphenols and whole‑grain fiber curtails inflammation and oxidative DNA damage, supporting a younger epigenetic profile. Adequate sleep (7–9 h/night) stabilizes DNA methylation of circadian and metabolic genes, protects telomere length and reduces hs‑CRP. Mind‑body stress reduction (mindfulness, breathing, social connection) lowers cortisol‑driven epigenetic inflammation,23aginghetic% and enhancing immune surveillance. Finally, abstaining from smoking, limiting alcohol, maintaining a healthy weight and staying mentally active complete the anti‑aging portfolio.
9 powerful ways to reverse aging – Regular aerobic exercise (≥150 min/week) improves VO₂ max, mitochondrial biogenesis and lowers epigenetic age by 1–2 years. Adding resistance training (2 sessions/week) preserves muscle mass, bone density and visceral fat, further reducing biological age markers. A Mediterranean‑style diet rich in omega‑3s, polyphenols and whole‑grain fiber curtails inflammation and oxidative DNA damage, supporting a younger epigenetic profile. Adequate sleep (7–9 h/night) stabilizes DNA methylation of circadian and metabolic genes, protects telomere length and reduces hs‑CRP. Mind‑body stress reduction (mindfulness, breathing, social connection) lowers cortisol‑driven epigenetic inflammation,23aginghetic% and enhancing immune surveillance. Finally, abstaining from smoking, limiting alcohol, maintaining a healthy weight and staying mentally active complete the anti‑aging portfolio.
How to reverse aging at 40 – Prioritize a balanced regimen of strength work (≥2 × week) and moderate cardio (≥150 min/week) to sustain lean mass and cardiovascular health. Adopt a plant‑forward, omega‑3‑rich diet while limiting processed sugars and saturated fats. Ensure 7–9 h of restorative sleep in a dark, cool environment and practice daily mindfulness or yoga. Schedule comprehensive biomarker panels (lipids, glucose, hs‑CRP, DNA‑methylation clocks) with a longevity‑focused clinician to personalize interventions and monitor progress.
The #1 mistake that ages you faster – Chronic insufficient, poor‑quality sleep is the single most damaging habit. Sleep deprivation disrupts DNA repair, elevates cortisol, accelerates telomere shortening and fuels systemic inflammation, collectively advancing biological age by several years (Biological Psychiatry 2020). Prioritizing uninterrupted, restorative sleep is the most effective single action to decelerate the aging clock.
How to delay the aging process – Combine regular aerobic and resistance exercise with a nutrient‑dense, anti‑inflammatory diet. Protect sleep, manage stress through meditation or breathing techniques, and undergo periodic biological‑age assessments (e.g., DunedinPACE, Horvath clock) to track and refine interventions. These evidence‑based strategies collectively preserve cellular energy, immune competence and healthspan.
Targeted Supplements and Nutrients
Supplement Summary
| Supplement | Typical Dose | Main Action | Reported Biological‑Age Effect |
|---|---|---|---|
| Vitamin D | 2,000 IU/day | Maintains telomere length, anti‑inflammatory | ~3 yr slowed cellular aging |
| NAD⁺ Precursors (NR/NMN) | 250‑500 mg/day | Boosts NAD⁺, improves mitochondrial efficiency | 1–2 yr reduction (12‑wk trial) |
| Omega‑3 Fatty Acids | 1‑2 g EPA/DHA/day | Anti‑inflammatory, membrane integrity | 0.2‑0.3 clock clock change (~3‑4 mo) |
| Senolytics (Dasatinib + Quercetin) | Clinical trial dosing | Clears senescent cells, ↓ SASP | Early data: functional gains, age ↓ ↓ |
| Met Metformin | 500‑1,000 mg/day (TAME trial) | Improves metabolic health, ↓ epigenetic age | ~1.5 yr reduction |
 Vitamin D is the vitamin that could help turn back the clock on biological aging. Trials in more than 900 adults showed that daily 2,000 IU supplementation preserved telomere length, preventing roughly 140 base‑pair losses per year – equivalent to up to three years of slowed cellular aging. Maintaining adequate vitamin D through sunlight, diet or supplements may therefore support healthier, longer lives.
Vitamin D is the vitamin that could help turn back the clock on biological aging. Trials in more than 900 adults showed that daily 2,000 IU supplementation preserved telomere length, preventing roughly 140 base‑pair losses per year – equivalent to up to three years of slowed cellular aging. Maintaining adequate vitamin D through sunlight, diet or supplements may therefore support healthier, longer lives.
NAD⁺ precursors – NAD⁺ boosters such as nicotinamide riboside or nicotinamide mononucleotide increase cellular NAD⁺ pools, improve mitochondrial efficiency, and have been shown to reduce epigenetic age by 1–2 years in 12‑week trials, while enhancing muscle strength and reducing fatigue.
Omega‑3 fatty acids – Consuming omega‑3‑rich foods (fatty fish, flaxseed, walnuts) attenuates chronic inflammation, supports membrane integrity, and modestly slows DNA‑methylation clocks (≈0.2‑0.3 units) over three years, translating into a 3‑4‑month reduction in biological age.
Senolytics and metformin – Senolytic combinations (dasatinib + quercetin) clear senescent cells, lower SASP cytokines, and improve physical function. Metformin, tested in the TAME trial, lowers epigenetic age by ~1.5 years and reduces cardiovascular events, suggesting a synergistic role in health‑span extension.
Which foods reduce biological age? Whole‑food, plant‑forward diets rich in antioxidants (berries, green tea, olive oil) and polyphenols, combined with omega‑3 sources and adequate vitamin D, consistently lower biological‑age markers.
Anti‑aging tech – Emerging approaches such as partial cellular reprogramming, gene‑therapy targeting PGD₂‑DP1 signaling, and NLRP3 inflammasome inhibition aim to reset age‑related molecular pathways, complementing nutritional strategies for a comprehensive anti‑aging regimen.
BioAge Labs – From Science to Therapies
BioAge Labs Pipeline
| Candidate | Target / Mechanism | Development Stage | Expected Milestones |
|---|---|---|---|
| BGE‑102 | NLRP3 inflammasome inhibitor | IND submission Q4 2026, Phase 1b/2a mid‑2026 | First human data on inflammation‑driven aging |
| APJ Agonist (Apelin receptor) | Mimics exercise‑derived peptide effects | IND filing late 2026 | Phase 1 trial start 2027 |
| Additional Multi‑omics‑driven therapeutics | Various hallmarks (mitochondrial, vascular) | Early discovery | Ongoing pre‑clinical validation |
 BioAge Labs, Inc. (NASDAQ: BIOA) is a California‑based biopharmaceutical company that applies a human‑first, multi‑omics discovery platform to pinpoint molecular drivers of metabolic aging. By integrating longitudinal clinical data, genomics, proteomics, and metabolomics, the platform isolates targets that link inflammation, mitochondrial dysfunction, and vascular health to accelerated biological age.
BioAge Labs, Inc. (NASDAQ: BIOA) is a California‑based biopharmaceutical company that applies a human‑first, multi‑omics discovery platform to pinpoint molecular drivers of metabolic aging. By integrating longitudinal clinical data, genomics, proteomics, and metabolomics, the platform isolates targets that link inflammation, mitochondrial dysfunction, and vascular health to accelerated biological age.
The company’s lead candidates focus on mimicking the systemic benefits of exercise. BGE‑102 is a novel NLRP3 inflammasome inhibitor designed to blunt chronic inflammation, improve endothelial function, and thereby slow metabolic aging. An IND for BGE‑102 is slated for submission by the end of 2026, with Phase 1b/2a trials expected to begin mid‑2026. In parallel, BioAge is developing an APJ (apelin) receptor agonist that reproduces the weight‑loss and muscle‑preserving effects of the exercise‑derived peptide apelin. The APJ program is projected to enter IND status by late 2026.
BioAge went public in early 2026, raising $115 million in a NASDAQ offering. Shares trade around $16 per share, giving a market cap near $672 million. Analysts see upside potential, citing the company’s extensive cash runway ($285 million) and the growing demand for therapies that target the biology of aging rather than individual disease endpoints. Upcoming clinical milestones for BGE‑102 and the APJ agonist will be key indicators of BioAge’s ability to translate its discovery platform into market‑ready, health‑span‑extending medicines.
Practical Lifestyle Toolkit for Everyday Energy
Daily Energy Checklist
| Habit | Frequency | Benefit to Biological Age |
|---|---|---|
| Hydration | 8‑10 cups water/day | Supports cellular metabolism, skin elasticity |
| Mind‑Body Practice | 10 min breathing ×2/day or mindfulness | Lowers cortisol, improves sleep, epigenetic marks |
| Social Engagement | Daily interaction (friends, community) | Reduces inflammatory cytokines, boosts T‑cell function |
| Sleep Hygiene | 7‑9 h, dark, cool bedroom | Preserves telomere length, DNA repair |
| Physical Activity | 150 min cardio + 2× resistance/week | Mitochondrial biogenesis, muscle preservation |
 Hydration: Adequate fluid intake supports cellular metabolism, maintains blood pressure, and prevents the dehydration‑driven loss of skin elasticity that accelerates biological aging. Studies link low fluid intake to higher sodium, increased mortality, and a biological age that exceeds chronological age; drinking 8‑10 cups of water daily helps keep organ systems, protect brain health ( and sustain energy levels.
Hydration: Adequate fluid intake supports cellular metabolism, maintains blood pressure, and prevents the dehydration‑driven loss of skin elasticity that accelerates biological aging. Studies link low fluid intake to higher sodium, increased mortality, and a biological age that exceeds chronological age; drinking 8‑10 cups of water daily helps keep organ systems, protect brain health ( and sustain energy levels.
Mind‑body practices: Regular mindfulness, breathing exercises, and yoga lower cortisol, reduce chronic inflammation, and improve sleep quality—all of which slow epigenetic age acceleration. A brief 10‑minute breathing routine twice daily has shown to reverse stress‑induced epigenetic markers, while 7‑9 hours of adequate sleep preserves telomere length and supports immune surveillance.
Social engagement: Strong relationships buffer stress‑related epigenetic damage and are among the most robust predictors of longevity. Frequent interaction with friends or community groups reduces inflammatory cytokines, bolsters T‑cell function, and improves mood, all contributing to a younger biological age and higher daily vitality.
Q1: Water & Aging – Adequate hydration maintains optimal cellular function, supports skin barrier integrity, and lowers mortality risk, thereby slowing biological aging.
Q2: Bullet‑proof immune – Whole‑food, omega‑3‑rich diet, 150 min/week moderate exercise, 7‑9 h sleep, stress‑reduction practices, tobacco cessation, and up‑to‑date vaccinations together restore immune competence.
Q3: Energy for a 70‑year‑old – Medical screen for anemia, thyroid, sleep apnea; adopt light resistance and walking 3‑5 times/week; practice consistent sleep hygiene; eat a balanced, nutrient‑dense diet; stay hydrated; track fatigue patterns; engage in enjoyable social activities.
Letn
Physical HY De engaged lifestyle habits into a measurable reduction in biological age, fortified immunity, and sustained daily energy.
Mindset, Social Connection, and Cognitive Vitality
Cognitive & Social Strategies
| Activity | Frequency | Epigenetic / Cognitive Effect |
|---|---|---|
| Learning New Skills (language, instrument) | Weekly practice | ↑ gray‑matter volume, slower brain aging |
| Social Interaction | Daily | ↓ CXCL9/IL‑6, 23 % lower stroke risk |
| Mindfulness Meditation | 10‑20 min/day | Halves pro‑inflammatory epigenetic marks |
| Structured Journaling | 5‑10 min/day | Improves stress resilience, supports DNA repair |
 Learning new skills stimulates neuroplasticity and expands gray‑matter volume, which epigenetic studies link to slower brain aging and lower epigenetic‑age scores. Regular cognitive challenges—language study, musical instrument practice, or complex puzzles—activate pathways that promote DNA‑methylation changes supportive of neuronal repair and mitochondrial efficiency.
Learning new skills stimulates neuroplasticity and expands gray‑matter volume, which epigenetic studies link to slower brain aging and lower epigenetic‑age scores. Regular cognitive challenges—language study, musical instrument practice, or complex puzzles—activate pathways that promote DNA‑methylation changes supportive of neuronal repair and mitochondrial efficiency.
Strong social relationships act as a buffer against chronic stress, a known accelerator of telomere shortening and inflammatory signaling. Epidemiological data show that individuals with robust social networks exhibit up to a 23% lower risk of stroke and a measurable reduction in biological‑age markers, likely mediated by lower circulating CXCL9 and IL‑6 levels.
Stress‑reduction techniques such as mindfulness meditation, structured breathing, and journaling attenuate cortisol‑driven epigenetic alterations, restoring immune competence and improving sleep architecture. These practices have been shown to halve pro‑inflammatory epigenetic marks in clinical cohorts.
People who have reversed aging – Entrepreneur Bryan Johnson’s $2 million “Project Blueprint” regimen—including vegan nutrition, daily exercise, metformin, lycopene, turmeric, lithium, and intensive monitoring—reported a five‑year reduction in biological age, with organ‑specific markers aligning with younger norms. Harvard Sinclair labs have documented modest epigenetic‑age improvements via plant‑based diets, caloric restriction, and fasting, while pre‑clinical Yamanaka factor gene‑therapy reverses epigenetic signatures in mice, illustrating a future pathway for human rejuvenation.
What is the #1 predictor of longevity? – Objective physical activity, captured by accelerometer‑derived movement patterns, outperforms chronological age, smoking status, and chronic disease burden in predicting all‑cause mortality. Even modest increases in daily movement significantly extend lifespan across age groups.
What diet reverses biological age? – A very‑low‑calorie ketogenic diet (VLCKD) has been shown to roll back obesity‑driven epigenetic aging by more than six years, demonstrating that metabolic re‑programming through strict carbohydrate restriction can reverse biological‑age acceleration.
Future Horizons: Emerging Therapies and Research
Emerging Anti‑Aging Interventions
| Therapy | Mechanism | Clinical Stage |
|---|---|---|
| Senolytics (Dasatinib + Quercetin, Fisetin) | Clear senescent cells | Early human trials (Phase 1/2) |
| Metformin (TAME) | Improves metabolic pathways, ↓ epigenetic age | Phase 3 completed, ongoing follow‑up |
| NAD⁺ Boosters (NR/NMN) | Replenish NAD⁺ pools | 12‑wk trials, Phase 2 ongoing |
| Partial Cellular Reprogramming (Yamanaka factors) | Reset epigenetic clocks without loss of identity | Pre‑clinical, early IND planning |
| Gene‑Therapy (TOR pathway, PGD₂‑DP1) | Modulate longevity pathways | Pre‑clinical to early Phase 1 |
| Rapamycin (TOR inhibitor) | Extends lifespan in models | Phase 2 trials for age‑related outcomes |
 New longevity research – Recent studies underscore the promise of senolytic agents that selectively clear senescent cells, with dasatinib‑quercetin and fisetin already showing health‑span gains in animal models and early human trials. Metformin and NAD⁺ precursors further lower epigenetic age by improving mitochondrial efficiency and reducing inflammation. Partial cellular reprogramming using transient Yamanaka‑factor expression has been demonstrated to restore youthful gene expression without erasing cell identity, offering a reversible route to tissue rejuvenation. Advanced epigenetic clocks such as DunedinPACE now provide precise, longitudinal measures of biological age, enabling clinicians to monitor the impact of these interventions in real time.
New longevity research – Recent studies underscore the promise of senolytic agents that selectively clear senescent cells, with dasatinib‑quercetin and fisetin already showing health‑span gains in animal models and early human trials. Metformin and NAD⁺ precursors further lower epigenetic age by improving mitochondrial efficiency and reducing inflammation. Partial cellular reprogramming using transient Yamanaka‑factor expression has been demonstrated to restore youthful gene expression without erasing cell identity, offering a reversible route to tissue rejuvenation. Advanced epigenetic clocks such as DunedinPACE now provide precise, longitudinal measures of biological age, enabling clinicians to monitor the impact of these interventions in real time.
Anti‑aging steps – A proactive, personalized regimen remains foundational. A nutrient‑dense, Mediterranean‑style diet rich in polyphenols, omega‑3s, and folate supports DNA‑methylation pathways; regular aerobic plus resistance exercise improves mitochondrial biogenesis and muscle quality; and 7‑9 hours of restorative sleep preserves telomere length and reduces inflammatory cytokines. Targeted supplementation (vitamin D, coenzyme Q10, and probiotic strains) should be guided by lab‑based biological‑age panels. Routine screenings and organ‑specific biomarker panels enable early detection of metabolic, cardiovascular, or immune dysregulation, allowing timely preventive or therapeutic action.
Anti‑aging tech – Cutting‑edge biotechnology is moving from concept to clinic. Partial epigenetic reprogramming, being evaluated by Altos Labs and Clock.bio, aims to reset cellular clocks while maintaining identity. Gene‑therapy delivery of longevity‑associated variants and pharmacologic modulation of the TOR pathway (e.g., rapamycin) are advancing through early‑phase trials. iPSC‑derived “self‑rejuvenation” cycles and organ‑on‑a‑chip platforms provide scalable models for testing interventions that could ultimately reverse age‑related decline and extend healthspan.
Putting It All Together – Your Personalized Longevity Plan
7 Ways to Slow Your Biological Clock
| # | Action | Core Reason |
|---|---|---|
| 1 | Anti‑inflammatory, alkaline‑rich diet (raw veg, fruits) | Reduces oxidative DNA damage |
| 2 | Limit refined carbs, saturated & trans fats | Lowers chronic inflammation |
| 3 | Prioritize omega‑3‑9 fats (fish, nuts, olive oil) | Improves membrane health & methylation |
| 4 | 12‑hour intermittent fasting 3‑4×/week | Aligns circadian methylation |
| 5 | Stay hydrated (8‑10 cups water) | Supports cellular metabolism |
| 6 | Quit smoking & limit alcohol | Prevents telomere shortening |
| 7 | Combine aerobic, strength, flexibility exercise (≈1 min per year of age daily) | Maintains mitochondrial function |
5 P’s to Avoid for Longevity
| P | Why It’s Harmful |
|---|---|
| Pizza | High‑calorie, refined carbs, pro‑inflammatory fats |
| Pasta | Refined grains → spikes insulin, glycation |
| Protein‑rich meats & dairy | Saturated fats, possible methionine excess |
| Potatoes | High glycemic index, inflammation spikes |
| Pane (bread) | Refined flour, low nutrient density |
 Reducing biological age means lowering the physiological “age” of your body so that it functions more like a younger person, reflected in improved DNA‑methylation patterns, reduced inflammation, and stronger organ function.
Reducing biological age means lowering the physiological “age” of your body so that it functions more like a younger person, reflected in improved DNA‑methylation patterns, reduced inflammation, and stronger organ function.
What diet reverses biological age? A ketogenic, very‑low‑calorie diet can roll back obesity‑linked epigenetic aging by over six years, while a fully vegan, plant‑based diet has shown an 0.63‑year reduction in epigenetic age after eight weeks, likely due to abundant epinutrients that support methylation.
Seven ways to slow your biological clock: adopt an anti‑inflammatory, alkaline‑rich diet rich in raw vegetables and fruits; limit refined carbs, saturated and trans fats; prioritize omega‑3‑9 fats; practice 12‑hour intermittent fasting 3‑4 times weekly; stay hydrated; quit smoking; combine aerobic, strength, and flexibility exercise (≈1 minute per year of age daily); and use mindfulness or personalized supplementation to curb stress.
What are the 5 P’s to avoid for longevity? Pizza, pasta, protein‑rich meats and dairy, potatoes, and pane (bread) are high‑calorie, inflammation‑driving foods that undermine healthy aging; swapping them for vegetables, nuts, legumes, and healthy fats aligns with a Mediterranean‑style diet that supports a lower biological age.
Bottom Line: Younger Biological Age = Greater Vitality
A lower biological age reflects a healthier cellular environment and translates directly into higher daily energy, stronger immune resilience, and a longer health‑span. Interventions that slow epigenetic aging—such as calorie‑restriction, regular aerobic‑plus‑resistance exercise, Mediterranean‑style nutrition, adequate sleep, and stress‑reduction—have been shown to improve mitochondrial ATP production, preserve telomere length, and reduce chronic inflammation (inflammaging). These physiological shifts boost naïve T‑cell output, enhance dendritic‑cell function, and lower pro‑inflammatory cytokines, thereby increasing resistance to infections and vaccines. Simultaneously, enhanced mitochondrial efficiency and reduced oxidative stress raise VO₂ max and overall stamina, delivering measurable gains in vitality. By consistently measuring biological age with epigenetic clocks (e.g., DunedinPACE) or multi‑omics panels, clinicians can personalize preventive strategies that compress morbidity, extending the years lived in robust health.
