Introduction
Autologous stem cell therapy exploits a patient’s own bone‑marrow or adipose‑derived mesenchymal stem cells (MSCs) to repair damaged musculoskeletal and hematopoietic tissues without provoking immune rejection. The cells are harvested, minimally processed under Good Manufacturing Practice conditions, and re‑injected into the target area under imaging guidance, where they secrete anti‑inflammatory cytokines, growth factors, and extracellular matrix components that stimulate angiogenesis, cartilage regeneration, and neural repair. This personalized regenerative approach aligns with contemporary strategies for extending healthspan by targeting the cellular mechanisms of aging and disease. The present success story illustrates how a multidisciplinary care model—combining a positive mindset, balanced nutrition, structured exercise, and a strong support network—can amplify the therapeutic benefits of an autologous stem cell transplant, enabling rapid restoration of mobility and quality of life after intensive oncology treatment.
Real‑World Success: From Cancer Recovery to Everyday Activity
Key Clinical Outcomes for Autologous Stem‑Cell Transplant (ASCT) in Multiple Myeloma
| Metric | Value | Notes |
|---|---|---|
| Partial remission rate | 80‑90 % | Typically achieved after high‑dose melphalan conditioning |
| 5‑year overall survival | 60‑70 % | Higher when patients enter transplant in deep remission and have favorable cytogenetics |
| Transplant‑related mortality | 2‑3 % | Modern supportive care reduces early deaths |
| Neutrophil engraftment | Day 10‑14 | Rapid recovery of innate immunity |
| Full immune competence | 3‑6 months | Varies with age and baseline health |
| Return to work/sport (median) | 12 months | Many resume full‑time work and regular exercise by 1 year |
These data illustrate why ASCT remains a cornerstone of curative intent for fit myeloma patients.
 Autologous stem‑cell transplantation (ASCT) delivers high‑dose chemotherapy while rescuing the patient’s own marrow, creating a powerful platform for durable remission in hematologic cancers. In multiple myeloma, ASCT consistently yields 80‑90 % partial remission rates and 5‑year overall survival of 60‑70 %, especially when patients enter transplant in deep remission and have favorable cytogenetics.
Autologous stem‑cell transplantation (ASCT) delivers high‑dose chemotherapy while rescuing the patient’s own marrow, creating a powerful platform for durable remission in hematologic cancers. In multiple myeloma, ASCT consistently yields 80‑90 % partial remission rates and 5‑year overall survival of 60‑70 %, especially when patients enter transplant in deep remission and have favorable cytogenetics.
Stem cell transplant for multiple myeloma recovery – After the transplant, blood counts rebound over 2‑4 weeks; patients experience transient fatigue, neutropenia, and thrombocytopenia, requiring prophylactic antibiotics, vigilant monitoring, nutrition, rest, and gradual exercise. Most feel markedly better after two weeks, but full immune competence may take several months.
Blood count normalization – High‑dose melphalan (200 mg/m²) is the standard conditioning regimen, with dose reductions for renal impairment. Supportive care includes hydration, antiemetics, and transfusion support. Modern protocols report transplant‑related mortality of only 2‑3 % and rapid engraftment of neutrophils by day 10‑14.
Return to work and sport – By one year post‑transplant, many patients, like Ian, have normalized blood counts, resume full‑time work, and engage in regular park runs and tennis, demonstrating restored mobility and energy. Early physical activity, guided by physiotherapy, accelerates functional recovery and preserves musculoskeletal health.
Is ASCT worth it? – For fit myeloma patients, ASCT offers deeper, longer remissions and improved quality of life, though overall survival now converges with novel‑agent regimens. Decisions should balance short‑term risks (infection, organ strain) against personal health goals.
Life expectancy after stem cell transplant for multiple myeloma – Contemporary data show 93 % survival at one year and 77 % at three years, with many achieving 5‑7‑year remissions when combined with modern induction, maintenance, and immunotherapy.
Autologous stem cell transplant success rate – Success is linked to younger age (<65 years), favorable disease biology, and deep pre‑transplant remission, making ASCT a cornerstone of curative intent in selected patients.
Athlete and Celebrity Transformations
Selected Athlete & Celebrity Stem‑Cell Cases
| Individual | Field | Stem‑cell therapy (type) | Reported benefit |
|---|---|---|---|
| George Kittle | NFL (tight end) | Bone‑marrow‑derived MSC injection + PRP | Dramatic reduction in tendon/joint pain, resumed training within weeks |
| T.J. Dillashaw | MMA champion | Autologous bone‑marrow MSCs | Accelerated recovery from joint injury, back performance |
| Cristiano Ronaldo | Soccer | Autologous MSCs | Decreased joint discomfort, quicker return to full training |
| Hutton Gibson (father of Mel Gibson) | Celebrity | Hip MSC procedure | Restored mobility and reduced pain |
| Bryan Johnson | Longevity influencer | Mesenchymal stem cells (systemic) | Anti‑aging effects, improved joint health |
Clinical studies corroborate these anecdotal reports, showing cartilage regeneration and pain reduction within 3‑12 months.
 High‑profile athletes and celebrities are increasingly turning to autologous stem‑cell therapies to accelerate recovery and extend performance longevity. Professional athletes such as NFL tight end George Kittle, MMA champion T.J. Dillashaw, and soccer star Cristiano Ronaldo have reported dramatic reductions in tendon and joint pain after receiving bone‑marrow‑derived stem‑cell injections, often combined with platelet‑rich plasma, allowing them to resume training within weeks (see multiple success‑story reports). Celebrities including Mel Gibson’s father Hutton Gibson, who cited a stem‑cell hip procedure for restored mobility, and longevity influencer Bryan Johnson, who uses mesenchymal stem cells for anti‑aging, further illustrate mainstream endorsement. These cases underscore the therapeutic potential of autologous stem cells for musculoskeletal repair, as clinical data show cartilage regeneration, pain reduction, and improved functional scores within 3–12 months. While the NFL does not ban stem‑cell treatments that are not performance‑enhancing substances, athletes must comply with league anti‑doping policies. Emerging evidence from regenerative clinics (e.g., ThriveMD, ID Clinic Bangkok confirms that stem‑cell injections can safely restore mobility after severe injuries, offering a viable, minimally invasive alternative to surgery for both elite performers and the general public.
High‑profile athletes and celebrities are increasingly turning to autologous stem‑cell therapies to accelerate recovery and extend performance longevity. Professional athletes such as NFL tight end George Kittle, MMA champion T.J. Dillashaw, and soccer star Cristiano Ronaldo have reported dramatic reductions in tendon and joint pain after receiving bone‑marrow‑derived stem‑cell injections, often combined with platelet‑rich plasma, allowing them to resume training within weeks (see multiple success‑story reports). Celebrities including Mel Gibson’s father Hutton Gibson, who cited a stem‑cell hip procedure for restored mobility, and longevity influencer Bryan Johnson, who uses mesenchymal stem cells for anti‑aging, further illustrate mainstream endorsement. These cases underscore the therapeutic potential of autologous stem cells for musculoskeletal repair, as clinical data show cartilage regeneration, pain reduction, and improved functional scores within 3–12 months. While the NFL does not ban stem‑cell treatments that are not performance‑enhancing substances, athletes must comply with league anti‑doping policies. Emerging evidence from regenerative clinics (e.g., ThriveMD, ID Clinic Bangkok confirms that stem‑cell injections can safely restore mobility after severe injuries, offering a viable, minimally invasive alternative to surgery for both elite performers and the general public.
Cost, Access, and Where to Seek Treatment
Typical Price Ranges & Geographic Options for Autologous Stem‑Cell Therapies
| Procedure | Typical US Cost (USD) | Example Locations |
|---|---|---|
| Full course (multiple joint injections) | $5,000‑$30,000 | California (Medical Institute of Healthy Aging), Texas (Riordan Medical Institute) |
| Single joint injection | $4,000‑$8,000 | Florida (Dr. Shiple’s Integrative Rehab Medicine) |
| Back‑pain multi‑level treatment | $3,000‑$7,000 per level | International clinics (Cayman Islands, Mexico) |
| Neck procedure | $4,000‑$12,000 | Same as above |
| Expanded‑cell / multi‑site protocols | $15,000‑$30,000+ | Specialized centers (ThriveMD, ID Clinic Bangkok) |
Insurance coverage is rare; most patients pay out‑of‑pocket or use financing options offered by clinics.
 Stem‑cell therapy costs vary widely. Most U.S. clinics charge $5,000‑$30,000 for a full course; a single joint injection often runs $4,000‑$8,000, while back‑pain treatments range $3,000‑$7,000 per level and neck procedures $4,000‑$12,000. Expanded‑cell products or multi‑site protocols can exceed $15,000‑$30,000. The Kellum Stem Cell Institute follows this market range and offers flexible financing, but does not publish a fixed price; patients should schedule a consultation for a tailored quote. Insurance rarely covers these experimental procedures, so out‑of‑pocket payment is typical.
Stem‑cell therapy costs vary widely. Most U.S. clinics charge $5,000‑$30,000 for a full course; a single joint injection often runs $4,000‑$8,000, while back‑pain treatments range $3,000‑$7,000 per level and neck procedures $4,000‑$12,000. Expanded‑cell products or multi‑site protocols can exceed $15,000‑$30,000. The Kellum Stem Cell Institute follows this market range and offers flexible financing, but does not publish a fixed price; patients should schedule a consultation for a tailored quote. Insurance rarely covers these experimental procedures, so out‑of‑pocket payment is typical.
Geographically, reputable options include California’s Medical Institute of Healthy Aging, Texas’ Riordan Medical Institute, Florida clinics like Dr. Shiple’s Integrative Rehab Medicine, and international destinations (Cayman Islands, Mexico) that may be 30‑50 % cheaper. Patients seeking treatment for arthritis, back, neck, or joint pain should look for FDA‑compliant, GMP‑certified facilities that use autologous bone‑marrow or adipose‑derived MSCs and provide image‑guided delivery.
When evaluating providers, review patient testimonials (e.g., Stem Cell Healing Institute) and verify that clinics adhere to the Arthritis Foundation’s “Unproven Stem Cell Intervention Patients’ Bill of Rights.” For personalized location matching, contact the Medical Institute of Healthy Aging, which can connect you with qualified California‑based physicians.
Safety, Regulations, and Clinical Evidence
Autologous vs. Allogeneic Stem‑Cell Transplant – Safety & Regulatory Snapshot
| Feature | Autologous (patient‑derived) | Allogeneic (donor‑derived) |
|---|---|---|
| FDA approval status (as of 2024) | No approved products for musculoskeletal use; hematopoietic products for blood disorders only | Same regulatory status for most MSC products; hematopoietic allogeneic transplants approved for specific indications |
| Matching requirement | None (patient’s own cells) | HLA‑matching required |
| GVHD risk | None | Present (up to 30 % in unrelated donors) |
| Treatment‑related mortality | <5 % (often 2‑3 % for ASCT) | Higher (5‑15 % depending on donor type) |
| Relapse risk | Higher (possible residual tumor cells) | Lower (graft‑versus‑malignancy effect) |
| Typical indications | Multiple myeloma, lymphoma, regenerative orthopedics (experimental) | Hematologic malignancies, bone‑marrow failure syndromes |
Regulatory agencies continue to evaluate IND pathways for orthopedic applications.
 Is stem cell therapy for back pain FDA‑approved?
No. The FDA has not approved any stem‑cell product for back pain or orthopedic conditions. Only hematopoietic stem cells from umbilical cord blood are cleared for blood‑disorder indications. All other regenerative‑medicine stem‑cell products are used outside clinical trials and may be illegal or unsafe without an IND.
Is stem cell therapy for back pain FDA‑approved?
No. The FDA has not approved any stem‑cell product for back pain or orthopedic conditions. Only hematopoietic stem cells from umbilical cord blood are cleared for blood‑disorder indications. All other regenerative‑medicine stem‑cell products are used outside clinical trials and may be illegal or unsafe without an IND.
Autologous vs allogeneic stem cell transplant Autologous transplants use the patient’s own cells, eliminating HLA‑matching, graft‑versus‑host disease (GVHD) and long‑term immunosuppression. They have lower treatment‑related mortality (<5 %) but higher relapse risk because the graft may contain residual tumor cells. Allogeneic transplants provide a tumor‑free graft and a graft‑versus‑malignancy effect, reducing recurrence, but carry higher risks of GVHD, graft failure, and mortality, especially with non‑identical donors.
Stem cell case study Matthew Farrow, diagnosed with Fanconi anaemia, received his sister’s umbilical‑cord blood transplant at age 5. The procedure restored hematopoiesis, allowing him to become a healthy adult. Similar cord‑blood protocols have shown motor‑function gains in cerebral palsy and autism trials, illustrating the long‑term benefits of personalized stem‑cell therapy.
Autologous stem cell therapy for osteoarthritis Autologous MSCs harvested from bone‑marrow aspirate concentrate or adipose tissue are injected intra‑articularly. Clinical trials (e.g., MILES) report pain reduction and functional improvement comparable to corticosteroids, with a favorable safety profile and no serious adverse events. Though experimental and not FDA‑approved for routine OA care, it offers a regenerative alternative to surgery pending further research.
Patient Stories Beyond the Clinic
Representative Patient Narratives & Outcomes
| Patient | Condition | Stem‑cell therapy | Key outcome |
|---|---|---|---|
| Ian | Multiple myeloma (post‑ASCT) | Autologous hematopoietic SCT | Normalized blood counts, returned to full‑time work, regular park runs & tennis after 1 year |
| Jocelyn | Chronic granulomatous disease | Haploidentical bone‑marrow transplant (sister’s donor) | Restored immune function, resumed school & sports |
| Andre | Knee & shoulder injuries | Autologous bone‑marrow MSC injections | Returned to gym training within 3 days, pain‑free range of motion |
| Chuck Dandridge | Genetic blood disorder (engineered sibling transplant) | Gene‑edited allogeneic SCT | Cancer‑free for nearly a decade |
| Multiple OA patients (clinical trial) | Knee osteoarthritis | Autologous MSC intra‑articular injection | Pain reduction ≥50 % and functional improvement sustained up to 24 months |
These stories highlight the potential for durable functional restoration across diverse disease contexts.
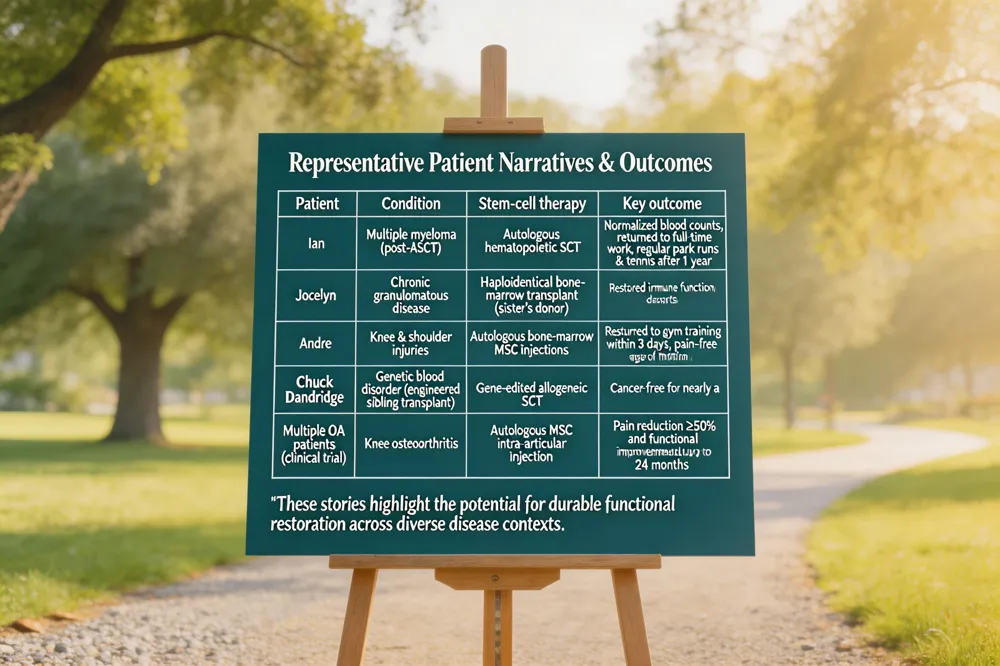 Stem‑cell patient stories illustrate how personalized regenerative approaches reshape daily life and long‑term health. Individual narratives such as Ian’s post‑autologous transplant recovery—normal blood counts, return to work, park runs and tennis—show how a positive mindset, balanced diet, exercise and strong support networks accelerate functional return. Pediatric cases like Jocelyn, who overcame chronic granulomatous disease after a haploidentical bone‑marrow transplant, highlight how donor‑expansion protocols restore immune competence, allowing children to resume school and sports. Successful stem‑cell cases extend to athletes; Andre returned to gym training within three days after autologous knee and shoulder injections, while high‑profile athletes report pain‑free performance after MSC therapy. Long‑term outcomes are evident in Chuck Dandridge’s genetically engineered sibling cell transplant, which has kept him cancer‑free for nearly a decade, and in studies showing sustained pain reduction and mobility gains for up to two years after autologous MSC injections for osteoarthritis and disc disease. These stories collectively demonstrate that autologous stem‑cell therapy—when combined with precise diagnostics, preventive lifestyle measures and tailored rehabilitation—can deliver durable functional restoration and quality‑of‑life improvements across a spectrum of hematologic, immunologic, and musculoskeletal conditions.
Stem‑cell patient stories illustrate how personalized regenerative approaches reshape daily life and long‑term health. Individual narratives such as Ian’s post‑autologous transplant recovery—normal blood counts, return to work, park runs and tennis—show how a positive mindset, balanced diet, exercise and strong support networks accelerate functional return. Pediatric cases like Jocelyn, who overcame chronic granulomatous disease after a haploidentical bone‑marrow transplant, highlight how donor‑expansion protocols restore immune competence, allowing children to resume school and sports. Successful stem‑cell cases extend to athletes; Andre returned to gym training within three days after autologous knee and shoulder injections, while high‑profile athletes report pain‑free performance after MSC therapy. Long‑term outcomes are evident in Chuck Dandridge’s genetically engineered sibling cell transplant, which has kept him cancer‑free for nearly a decade, and in studies showing sustained pain reduction and mobility gains for up to two years after autologous MSC injections for osteoarthritis and disc disease. These stories collectively demonstrate that autologous stem‑cell therapy—when combined with precise diagnostics, preventive lifestyle measures and tailored rehabilitation—can deliver durable functional restoration and quality‑of‑life improvements across a spectrum of hematologic, immunologic, and musculoskeletal conditions.
Conclusion
Key takeaways: Autologous stem cell approaches—whether as high‑dose hematopoietic transplants for hematologic cancers or localized mesenchymal injections for musculoskeletal injury—consistently restore blood counts, reduce pain, and improve functional mobility. Harvesting a patient’s own marrow or adipose tissue minimizes immune rejection, while emerging adjuncts such as PRP, platelet‑rich plasma, and lysosomal‑targeting therapies further enhance tissue repair. Future outlook: Ongoing research into lysosomal rejuvenation of blood‑forming stem cells, exosome‑rich placental products now permitted in Florida, and large‑scale MSC trials in frailty suggest broader applications for age‑related degeneration. As regulatory pathways solidify and long‑term safety data accumulate, personalized autologous stem cell regimens are poised to become a cornerstone of preventive longevity medicine.
