Why Early Detection Matters
Neurodegenerative diseases such as Alzheimer’s and Parkinson’s begin 10–20 years before any clinical signs, with soluble oligomeric proteins, neuroinflammation, and vascular breakdown occurring silently (Early detection of these prodromal changes—through fluid biomarkers (plasma p‑tau217, NfL, GFAP, Aβ42/40), neuro‑imaging (amyloid‑ and tau‑PET, high‑resolution MRI), or digital phenotyping (wearable gait, speech analytics)—enables initiation of disease‑modifying therapies while neuronal networks are still viable. Interventions applied at this stage, including anti‑amyloid antibodies, tau aggregation inhibitors, and non‑pharmacological strategies (aerobic exercise, Mediterranean diet, cognitive training), have shown the greatest impact on preserving cognition and delaying functional decline. Personalized longevity programs, such as those offered by the Medical Institute of Healthy Aging, integrate these biomarkers with genetic risk profiling (APOE ε4, polygenic scores) and continuous monitoring, allowing individualized preventive plans that target the specific pathogenic pathways active in each patient. Consequently, early, tailored intervention can transform neurodegenerative trajectories, improve quality of life, and extend healthspan.
Histology: The Microscopic Lens on Disease
Histology – Quick Reference Table
| Key Hallmarks | Regions Assessed | Clinical Utility | Validation Role |
|---|---|---|---|
| Amyloid‑β plaques, tau neurofibrillary tangles, α‑synuclein inclusions | Hippocampus, Frontal Cortex, Substantia Nigra | Early differential diagnosis (AD vs PD vs mixed) & enrollment in disease‑modifying trials | Gold‑standard for confirming fluid & imaging biomarkers (e.g., p‑tau181, PET tracers) |
| Quantification of burden, distribution, biochemical state | Same as above | Guides personalized treatment plans & monitors progression | Provides tissue‑level correlation for blood‑based assays (Aβ42/40, NfL) |
| Rapid neuropathological screening | Targeted brain regions | Enables timely preventive interventions | Refines and calibrates non‑invasive diagnostics |
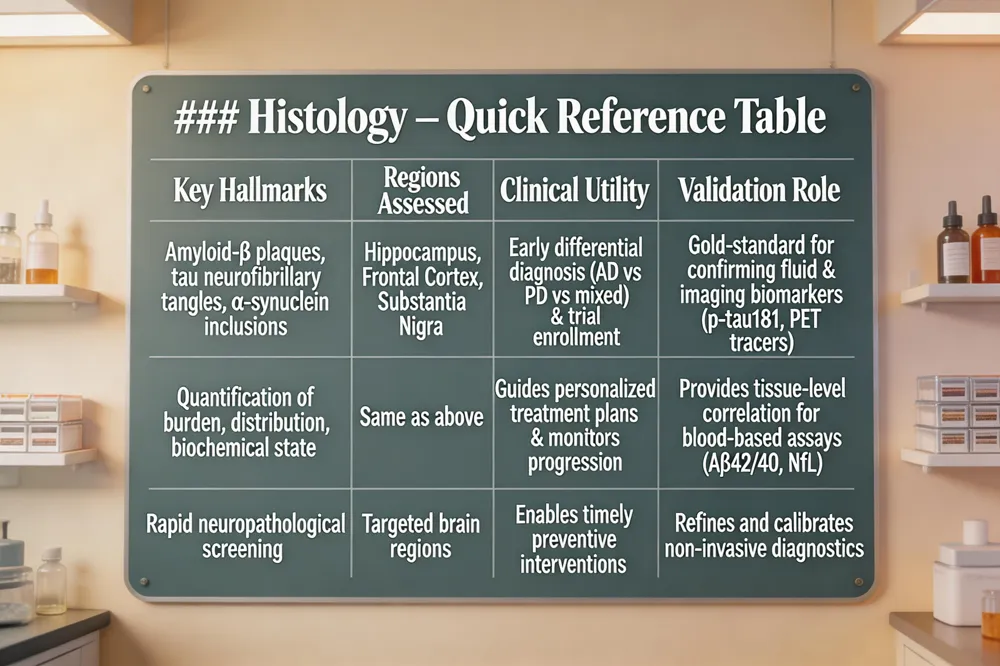 Histological examinations uncover the microscopic hallmarks of neurodegeneration—amyloid‑β plaques, tau neurofibrillary tangles, and α‑synuclein inclusions—often years before overt clinical symptoms arise. By quantifying the burden, distribution, and biochemical state of these aggregates, pathologists can differentiate Alzheimer's disease from Parkinson's disease and identify mixed or atypical pathologies that impact therapeutic decisions. Rapid neuropathological screening of key regions such as the hippocampus, frontal cortex, and substantia nigra enables early diagnosis, facilitating enrollment in disease‑modifying trials and timely preventive interventions. Moreover, histology validates and refines non‑invasive fluid and imaging biomarkers, ensuring that blood‑based assays (e.g., p‑tau181, p‑tau217, NfL) and PET tracers accurately reflect underlying tissue changes. Integrating histological insights into clinical workflows supports personalized treatment plans, monitors disease progression, and improves prognosis for individuals at risk of neurodegeneration.
Histological examinations uncover the microscopic hallmarks of neurodegeneration—amyloid‑β plaques, tau neurofibrillary tangles, and α‑synuclein inclusions—often years before overt clinical symptoms arise. By quantifying the burden, distribution, and biochemical state of these aggregates, pathologists can differentiate Alzheimer's disease from Parkinson's disease and identify mixed or atypical pathologies that impact therapeutic decisions. Rapid neuropathological screening of key regions such as the hippocampus, frontal cortex, and substantia nigra enables early diagnosis, facilitating enrollment in disease‑modifying trials and timely preventive interventions. Moreover, histology validates and refines non‑invasive fluid and imaging biomarkers, ensuring that blood‑based assays (e.g., p‑tau181, p‑tau217, NfL) and PET tracers accurately reflect underlying tissue changes. Integrating histological insights into clinical workflows supports personalized treatment plans, monitors disease progression, and improves prognosis for individuals at risk of neurodegeneration.
Core Alzheimer’s Biomarkers: The Biological Signature
Core AD Biomarkers – Summary Table
| Biomarker | Fluid Type | Diagnostic Value | Typical Ratio/Combination |
|---|---|---|---|
| Aβ42 (or Aβ42/Aβ40) | CSF (or plasma) | Low levels indicate amyloid deposition | Aβ42/Aβ40 ratio ↓ |
| Total tau (t‑tau) | CSF | Elevated in neurodegeneration | t‑tau/Aβ42 ↑ |
| Phosphorylated tau (p‑tau181, p‑tau217) | CSF (or plasma) | Highly specific for tau pathology | p‑tau/Aβ42 ↑ |
| Neurofilament light chain (NfL) | CSF or plasma | Marker of axonal injury, nonspecific neurodegeneration | — |
| Composite (p‑tau/Aβ42, t‑tau/Aβ42) | CSF | Improves specificity for AD vs non‑AD dementias | — |
| --- |
 Alzheimer’s disease (AD) is defined by the accumulation of amyloid‑β (Aβ) plaques and neurofibrillary tangles composed of hyper‑phosphorylated tau. The most reliable fluid biomarkers are low cerebrospinal‑fluid (CSF) Aβ42 (or a reduced Aβ42/Aβ40 ratio) and elevated CSF tau species – total tau (t‑tau) and phosphorylated tau (p‑tau, especially p‑tau181 and p‑tau217). These markers mirror amyloid‑PET and tau‑PET findings and become abnormal years before cognitive symptoms appear. Neurofilament light chain (NfL), measurable in CSF or plasma, rises with axonal injury and provides a sensitive indicator of neurodegeneration across AD and other disorders. Diagnostic accuracy is sharpened by combining ratios such as p‑tau/Aβ42 or t‑tau/Aβ42, which improve specificity for AD versus non‑AD dementias. Together, the Aβ‑tau axis and NfL form a biological signature that enables minimally invasive, early detection, risk stratification, and monitoring of therapeutic response in personalized longevity programs.
Alzheimer’s disease (AD) is defined by the accumulation of amyloid‑β (Aβ) plaques and neurofibrillary tangles composed of hyper‑phosphorylated tau. The most reliable fluid biomarkers are low cerebrospinal‑fluid (CSF) Aβ42 (or a reduced Aβ42/Aβ40 ratio) and elevated CSF tau species – total tau (t‑tau) and phosphorylated tau (p‑tau, especially p‑tau181 and p‑tau217). These markers mirror amyloid‑PET and tau‑PET findings and become abnormal years before cognitive symptoms appear. Neurofilament light chain (NfL), measurable in CSF or plasma, rises with axonal injury and provides a sensitive indicator of neurodegeneration across AD and other disorders. Diagnostic accuracy is sharpened by combining ratios such as p‑tau/Aβ42 or t‑tau/Aβ42, which improve specificity for AD versus non‑AD dementias. Together, the Aβ‑tau axis and NfL form a biological signature that enables minimally invasive, early detection, risk stratification, and monitoring of therapeutic response in personalized longevity programs.
Multimodal Early‑Detection Toolbox
Multimodal Early‑Detection Toolbox – Key Modalities
| Modality | Example | What it Detects | Clinical Stage |
|---|---|---|---|
| Neurocognitive testing | MoCA, MMSE | Subtle cognitive deficits | Screening |
| MRI (structural) | 3 T T1/T2 | Hippocampal atrophy, cortical thinning | Early detection |
| PET (amyloid & tau) | ^18F‑florbetapir, ^18F‑flortaucipir | Molecular plaque/tangle burden | Confirmatory |
| CSF biomarkers | Aβ42, p‑tau181, NfL | Direct CNS fluid signatures | Diagnostic |
| Plasma biomarkers | Aβ42/40, p‑tau217, NfL, GFAP | Minimally invasive disease signature | Screening/monitoring |
| Digital biomarkers | Wearable gait sensors, speech analysis apps | Motor & language changes in real‑time | Continuous monitoring |
| Point‑of‑care biosensors | GaN‑HEMT sensors for S100B, UCH‑L1, GFAP, Aβ | Ultra‑sensitive protein detection (fg) | Rapid bedside triage |
 Early detection of neurodegenerative diseases integrates clinical neurocognitive testing with advanced neuroimaging such as MRI and PET scans that reveal structural and molecular changes before symptoms appear. Fluid biomarkers measured in cerebrospinal fluid and blood—including **amyloid‑β, total and phosphorylated tau, Neurofilament light chain, and emerging micro‑RNA signatures—provide molecular evidence of disease pathology. Non‑invasive digital biomarkers derived from **wearable sensors, smartphone or tablet applications, and automated speech and gait analyses capture subtle behavioral and motor alterations in real time. Point‑of‑care biosensing platforms (e.g., GaN‑HEMT sensors for S100B, UCH‑L1, GFAP, and Aβ) enable ultra‑sensitive detection of protein biomarkers at femtogram concentrations. Together, these multimodal tools allow clinicians to identify at‑risk individuals years before overt clinical decline, facilitating timely, personalized interventions.
Early detection of neurodegenerative diseases integrates clinical neurocognitive testing with advanced neuroimaging such as MRI and PET scans that reveal structural and molecular changes before symptoms appear. Fluid biomarkers measured in cerebrospinal fluid and blood—including **amyloid‑β, total and phosphorylated tau, Neurofilament light chain, and emerging micro‑RNA signatures—provide molecular evidence of disease pathology. Non‑invasive digital biomarkers derived from **wearable sensors, smartphone or tablet applications, and automated speech and gait analyses capture subtle behavioral and motor alterations in real time. Point‑of‑care biosensing platforms (e.g., GaN‑HEMT sensors for S100B, UCH‑L1, GFAP, and Aβ) enable ultra‑sensitive detection of protein biomarkers at femtogram concentrations. Together, these multimodal tools allow clinicians to identify at‑risk individuals years before overt clinical decline, facilitating timely, personalized interventions.
Gene Therapy: Re‑Engineering the Brain
Gene Therapy Landscape – Core Elements
| Vector/Platform | Target Disease | Gene/Approach | Clinical Status |
|---|---|---|---|
| AAV (intrathecal/IV) | Parkinson’s disease | GDNF or aromatic L‑amino acid decarboxylase delivery | Phase I/II trials |
| AAV | Huntington’s disease | anti‑HTT RNAi | Phase I trial |
| AAV | ALS (SOD1, C9orf72) | Gene‑silencing via RNAi/ASO | Phase I/II trials |
| Non‑viral nanoparticles | Various neurodegenerations | CRISPR‑Cas9 or base editing for allele correction | Pre‑clinical/early‑phase |
| Lipid‑based carriers | Broad CNS targets | Delivery of CRISPR components or antisense oligos | Pre‑clinical |
 Gene therapy is being explored as a disease‑modifying strategy for neurodegenerative disorders by delivering neuroprotective genes, silencing toxic mutant proteins, or re‑wiring pathogenic pathways directly within the central nervous system. The most common delivery platform is adeno‑associated virus (AAV) vectors, which can be administered intrathecally or intravenously and have been engineered for efficient blood‑brain barrier crossing and widespread neuronal transduction. Emerging non‑viral nanoparticles and lipid‑based carriers also show promise for targeted delivery. Genome‑editing tools such as CRISPR/Cas9, base editors, RNA‑interference, and antisense oligonucleotides are employed to correct or knock down disease‑causing alleles, offering the potential for permanent modification of disease trajectories. Ongoing clinical trials include AAV‑mediated delivery of GDNF or aromatic L‑amino acid decarboxylase for Parkinson’s disease, anti‑HTT RNAi for Huntington’s disease, and SOD1 or C9orf72‑targeted approaches for amyotrophic lateral sclerosis. While challenges remain—including immune responses, vector size constraints, and precise cell‑type targeting—the rapid advances in vector engineering and genome‑editing technologies are bringing durable, personalized gene‑based interventions closer to clinical reality for aging patients.
Gene therapy is being explored as a disease‑modifying strategy for neurodegenerative disorders by delivering neuroprotective genes, silencing toxic mutant proteins, or re‑wiring pathogenic pathways directly within the central nervous system. The most common delivery platform is adeno‑associated virus (AAV) vectors, which can be administered intrathecally or intravenously and have been engineered for efficient blood‑brain barrier crossing and widespread neuronal transduction. Emerging non‑viral nanoparticles and lipid‑based carriers also show promise for targeted delivery. Genome‑editing tools such as CRISPR/Cas9, base editors, RNA‑interference, and antisense oligonucleotides are employed to correct or knock down disease‑causing alleles, offering the potential for permanent modification of disease trajectories. Ongoing clinical trials include AAV‑mediated delivery of GDNF or aromatic L‑amino acid decarboxylase for Parkinson’s disease, anti‑HTT RNAi for Huntington’s disease, and SOD1 or C9orf72‑targeted approaches for amyotrophic lateral sclerosis. While challenges remain—including immune responses, vector size constraints, and precise cell‑type targeting—the rapid advances in vector engineering and genome‑editing technologies are bringing durable, personalized gene‑based interventions closer to clinical reality for aging patients.
Blood‑Based Alzheimer’s Biomarkers: From Bench to Clinic
Blood‑Based AD Biomarkers – Clinical Implementation
| Biomarker | Assay Type | Clinical Sensitivity | Clinical Specificity |
|---|---|---|---|
| Plasma p‑tau217 | FDA‑cleared (Fujirebio) | ≥90 % for amyloid pathology | ≥75 % |
| Plasma p‑tau181 | FDA‑cleared (Roche Elecsys) | ≥85 % | ≥70 % |
| Plasma Aβ42/40 ratio | Research‑grade ELISA | 80‑90 % | 70‑80 % |
| NfL (plasma) | Research‑grade Simoa | Sensitive to neurodegeneration across disorders | Moderate specificity |
| GFAP (plasma) | Research‑grade | Early astrocytic response detection | Moderate specificity |
 Plasma Aβ42/40 ratio and phosphorylated tau isoforms (p‑tau181, p‑tau217) have emerged as the core blood‑based biomarkers for Alzheimer’s disease. The Aβ42/40 ratio mirrors cerebrospinal‑fluid reductions and predicts amyloid PET positivity, while p‑tau181 and especially p‑tau217 closely track tau pathology and correlate with cognitive decline. Regulatory milestones accelerated translation: the FDA cleared the Fujirebio plasma p‑tau217 assay and Roche Elecsys plasma p‑tau181 test for diagnostic use in individuals with mild cognitive impairment, establishing the first FDA‑cleared blood tests for AD. In large community cohorts, these biomarkers demonstrate robust performance—p‑tau217 and p‑tau181 achieve area‑under‑the‑curve (AUC) values of 0.88–0.96 for detecting amyloid burden, and when combined with NfL or GFAP, predictive accuracy for incident AD dementia exceeds 80 %. Such data support a screening paradigm where minimally invasive blood assays stratify risk, guide further imaging or CSF confirmation, and enable early, personalized interventions to preserve healthspan.
Plasma Aβ42/40 ratio and phosphorylated tau isoforms (p‑tau181, p‑tau217) have emerged as the core blood‑based biomarkers for Alzheimer’s disease. The Aβ42/40 ratio mirrors cerebrospinal‑fluid reductions and predicts amyloid PET positivity, while p‑tau181 and especially p‑tau217 closely track tau pathology and correlate with cognitive decline. Regulatory milestones accelerated translation: the FDA cleared the Fujirebio plasma p‑tau217 assay and Roche Elecsys plasma p‑tau181 test for diagnostic use in individuals with mild cognitive impairment, establishing the first FDA‑cleared blood tests for AD. In large community cohorts, these biomarkers demonstrate robust performance—p‑tau217 and p‑tau181 achieve area‑under‑the‑curve (AUC) values of 0.88–0.96 for detecting amyloid burden, and when combined with NfL or GFAP, predictive accuracy for incident AD dementia exceeds 80 %. Such data support a screening paradigm where minimally invasive blood assays stratify risk, guide further imaging or CSF confirmation, and enable early, personalized interventions to preserve healthspan.
Lifestyle as a Neuroprotective Prescription
Lifestyle Prescription – Core Components
| Intervention | Primary Mechanism | Evidence Highlights | Recommended Dose/Frequency |
|---|---|---|---|
| Mediterranean‑style diet | Antioxidant & anti‑inflammatory nutrients (omega‑3, flavonoids) | Reduces AD risk, lowers inflammatory markers | Daily meals rich in vegetables, fish, olive oil |
| Aerobic & resistance exercise | Increases cerebral blood flow, neurogenesis, insulin sensitivity | Improves cognition, mitigates vascular injury | ≥150 min moderate‑intensity aerobic + 2 sessions resistance per week |
| Sleep hygiene & stress reduction (meditation, yoga) | Lowers cortisol, reduces chronic neuroinflammation | Correlates with lower GFAP/sTREM2 | 7‑9 h sleep/night; 10‑20 min daily mindfulness |
| Smoking cessation & moderate alcohol | Prevents vascular injury, toxic metabolite buildup | Decreases dementia incidence | No smoking; ≤1 drink/day for women, ≤2 for men |
| Cognitive & social engagement | Builds cognitive reserve, strengthens neural networks | Delays onset of AD & PD symptoms | Lifelong learning, group activities, volunteering |
 Adopting a Mediterranean‑style diet rich in omega‑3 fatty acids, antioxidants, flavonoids and low in saturated fats supplies neuroprotective nutrients that curb inflammation and oxidative stress, key drivers of neuronal loss (Solving neurodegeneration: common mechanisms and strategies). Regular aerobic exercise and resistance training increase cerebral blood flow, stimulate neurogenesis, and enhance synaptic plasticity while improving insulin sensitivity and mitochondrial function (Physical activity and lifestyle modifications in the treatment of ...). Sufficient, high‑quality sleep and stress‑reduction practices such as meditation or yoga lower cortisol and mitigate chronic neuroinflammation, protecting against glial activation markers like GFAP and sTREM2 (Decoding Neuroinflammation Biomarkers). Avoiding smoking, limiting excess alcohol, and maintaining a healthy weight further prevent vascular injury and toxic metabolite buildup that accelerate neurodegeneration. Finally, lifelong cognitive and social engagement builds cognitive reserve, strengthening neural networks and delaying onset of Alzheimer’s and Parkinson’s disease (Early detection of neurodegeneration and early Alzheimer's detection).
Adopting a Mediterranean‑style diet rich in omega‑3 fatty acids, antioxidants, flavonoids and low in saturated fats supplies neuroprotective nutrients that curb inflammation and oxidative stress, key drivers of neuronal loss (Solving neurodegeneration: common mechanisms and strategies). Regular aerobic exercise and resistance training increase cerebral blood flow, stimulate neurogenesis, and enhance synaptic plasticity while improving insulin sensitivity and mitochondrial function (Physical activity and lifestyle modifications in the treatment of ...). Sufficient, high‑quality sleep and stress‑reduction practices such as meditation or yoga lower cortisol and mitigate chronic neuroinflammation, protecting against glial activation markers like GFAP and sTREM2 (Decoding Neuroinflammation Biomarkers). Avoiding smoking, limiting excess alcohol, and maintaining a healthy weight further prevent vascular injury and toxic metabolite buildup that accelerate neurodegeneration. Finally, lifelong cognitive and social engagement builds cognitive reserve, strengthening neural networks and delaying onset of Alzheimer’s and Parkinson’s disease (Early detection of neurodegeneration and early Alzheimer's detection).
Blood Biomarkers for Dementia Diagnosis
Composite Blood Biomarker Panels – Diagnostic Workflow
| Panel Components | Diagnostic Metric | AUC (Amyloid PET) | Clinical Workflow |
|---|---|---|---|
| Aβ42/40 × p‑tau217 | Amyloid PET positivity prediction | ≈0.95 | Primary‑care screen → confirmatory CSF/PET if positive |
| Aβ42/40, p‑tau181, NfL, GFAP | Differentiation of AD vs frontotemporal & vascular dementias | 0.88–0.96 (AD) | Tiered: rapid panel → targeted imaging/CSF → therapeutic decision |
| p‑tau217 + NfL | Early axonal injury & tau pathology | 0.90 (AD) | Early risk stratification, monitor disease progression |
 Composite panels that combine plasma Aβ42/40 ratio, p‑tau181 and p‑tau217 (phosphorylated tau isoforms), NfL (neurofilament light chain) and GFAP (glial fibrillary acidic protein) offer the most robust diagnostic strategy. The amyloid‑tau ratio (Aβ42/40 × p‑tau217) accurately predicts amyloid PET positivity (AUC ≈ 0.95) and distinguishes Alzheimer’s disease from other dementias, while NfL and GFAP capture neuroaxonal injury and astrocytic activation common to frontotemporal dementias. Across subtypes, p‑tau217 shows highest specificity for AD, whereas NfL rises earliest in frontotemporal and vascular disorders; GFAP is especially sensitive to early astroglial response in mixed pathologies. Clinically, a tiered workflow is recommended: initial screening with a rapid blood‑based panel (Aβ42/40, p‑tau181/p‑tau217, NfL, GFAP) in primary‑care settings, followed by confirmatory CSF analysis or PET imaging for individuals with positive or equivocal results. This approach enables efficient triage, reduces reliance on invasive lumbar puncture, and facilitates early enrollment in disease‑modifying therapies.
Composite panels that combine plasma Aβ42/40 ratio, p‑tau181 and p‑tau217 (phosphorylated tau isoforms), NfL (neurofilament light chain) and GFAP (glial fibrillary acidic protein) offer the most robust diagnostic strategy. The amyloid‑tau ratio (Aβ42/40 × p‑tau217) accurately predicts amyloid PET positivity (AUC ≈ 0.95) and distinguishes Alzheimer’s disease from other dementias, while NfL and GFAP capture neuroaxonal injury and astrocytic activation common to frontotemporal dementias. Across subtypes, p‑tau217 shows highest specificity for AD, whereas NfL rises earliest in frontotemporal and vascular disorders; GFAP is especially sensitive to early astroglial response in mixed pathologies. Clinically, a tiered workflow is recommended: initial screening with a rapid blood‑based panel (Aβ42/40, p‑tau181/p‑tau217, NfL, GFAP) in primary‑care settings, followed by confirmatory CSF analysis or PET imaging for individuals with positive or equivocal results. This approach enables efficient triage, reduces reliance on invasive lumbar puncture, and facilitates early enrollment in disease‑modifying therapies.
Emerging Non‑Invasive Imaging and Digital Tools
Emerging Imaging & Digital Tools – Validation Snapshot
| Tool | Modality | Target Pathology | Validation Status |
|---|---|---|---|
| NIRF probes (BD‑Oligo, PTO‑29, F‑SLOH) | Near‑infrared fluorescence imaging | Soluble Aβ oligomers | In‑vivo mouse validation; pilot human studies |
| Optical Coherence Tomography (OCT) | Ophthalmic imaging | Retinal nerve‑fiber thinning (AD) | Correlates with CNS amyloid/tau; clinical trials |
| OCT‑angiography | Ophthalmic imaging | Microvascular alterations (PD, AD) | Emerging evidence, multicenter studies |
| AI‑driven speech analysis | Digital audio processing | Early language decline | >80 % accuracy in MCI vs healthy aging |
| AI‑driven gait analysis | Wearable sensors + ML | Motor slowdown, dual‑task deficits | >80 % accuracy in early neurodegeneration |
| Resting‑state EEG ML models | EEG signal processing | Network dysfunction, early AD | >80 % classification accuracy |
 Near‑infrared fluorescence (NIRF) imaging has emerged as a radiation‑free, cost‑effective method for detecting soluble amyloid‑β oligomers. Probes such as BD‑Oligo, PTO‑29 and the fluorinated cyanine F‑SLOH cross the blood‑brain barrier, fluoresce upon binding Aβ oligomers, and have been validated for in‑vivo imaging in APP/PS1 mouse brains, offering a potential bedside screening tool. Parallel advances in ophthalmic imaging—optical coherence tomography (OCT) and OCT‑angiography—provide a non‑invasive window onto neurodegeneration. Thinning of the retinal nerve‑fiber layer and microvascular alterations detected by these modalities correlate with early Alzheimer’s and Parkinson’s pathology, mirroring central nervous system changes before clinical symptoms appear. In the digital arena, AI‑driven analysis of speech patterns, gait kinematics, and resting‑state EEG is now capable of flagging subtle cognitive and motor decline. Machine‑learning models that integrate acoustic biomarkers, dual‑task gait metrics, and EEG band‑power reductions achieve >80 % accuracy in distinguishing mild cognitive impairment from healthy aging. Recent updates in Alzheimer’s disease biomarker research include ultra‑sensitive plasma assays for p‑tau217 and p‑tau181, which, when combined with Aβ42/40 ratio, neurofilament light chain, and glial fibrillary acidic protein, generate composite risk scores with ≥90 % sensitivity and ≥75 % specificity for amyloid pathology. Large‑scale validation studies (2024‑2025) across diverse cohorts have led to the Alzheimer’s Association’s first guideline recommending blood‑based tests as a triage tool, positioning these minimally invasive diagnostics as a cornerstone of personalized longevity programs.
Near‑infrared fluorescence (NIRF) imaging has emerged as a radiation‑free, cost‑effective method for detecting soluble amyloid‑β oligomers. Probes such as BD‑Oligo, PTO‑29 and the fluorinated cyanine F‑SLOH cross the blood‑brain barrier, fluoresce upon binding Aβ oligomers, and have been validated for in‑vivo imaging in APP/PS1 mouse brains, offering a potential bedside screening tool. Parallel advances in ophthalmic imaging—optical coherence tomography (OCT) and OCT‑angiography—provide a non‑invasive window onto neurodegeneration. Thinning of the retinal nerve‑fiber layer and microvascular alterations detected by these modalities correlate with early Alzheimer’s and Parkinson’s pathology, mirroring central nervous system changes before clinical symptoms appear. In the digital arena, AI‑driven analysis of speech patterns, gait kinematics, and resting‑state EEG is now capable of flagging subtle cognitive and motor decline. Machine‑learning models that integrate acoustic biomarkers, dual‑task gait metrics, and EEG band‑power reductions achieve >80 % accuracy in distinguishing mild cognitive impairment from healthy aging. Recent updates in Alzheimer’s disease biomarker research include ultra‑sensitive plasma assays for p‑tau217 and p‑tau181, which, when combined with Aβ42/40 ratio, neurofilament light chain, and glial fibrillary acidic protein, generate composite risk scores with ≥90 % sensitivity and ≥75 % specificity for amyloid pathology. Large‑scale validation studies (2024‑2025) across diverse cohorts have led to the Alzheimer’s Association’s first guideline recommending blood‑based tests as a triage tool, positioning these minimally invasive diagnostics as a cornerstone of personalized longevity programs.
From Detection to Proactive Longevity
Early detection of neurodegeneration now leverages a multimodal biomarker toolbox that includes ultra‑sensitive plasma assays (p‑tau181/217, NfL, Aβ42/40), CSF analyses, and FDA‑approved PET tracers for Aβ and tau. Non‑invasive fluid sources such as saliva, tears, and nasal secretions are emerging, while near‑infrared fluorescent probes and high‑resolution MRI provide radiation‑free imaging of oligomeric aggregates. The Medical Institute of Healthy Aging (MDIHA) integrates these data streams into a personalized monitoring platform that tracks longitudinal biomarker trajectories, genetic risk (APOE ε4, polygenic scores), and lifestyle metrics (wearable‑derived sleep, gait, and activity). Clinicians receive risk dashboards that trigger early interventions—targeted exercise, Mediterranean diet, and anti‑inflammatory nutraceuticals—before irreversible neuronal loss. Looking ahead, gene‑editing approaches (e.g., APOE4 silencing) and digital health tools (continuous EEG, smartphone‑based cognitive phenotyping) will expand the preventive armamentarium, enabling truly proactive longevity strategies that align molecular diagnostics with individualized therapeutic regimens.
